Trial Insights
The Trial Insights provides the following information for each Clinical Trials. (Across all countries and for each country):
- Trial Protocol Information
- Trial Milestones
- Investigational Site Inventory
- Investigational Site In-Transit Shipments
- Subject Screening and Enrollment
- Upcoming Site Visits
NOTE: On navigation to Trial Insights tab if you cannot see correct data / if you want to reset the selection on the dashboard then click on
- Reset/Clear selection button in slicers pane.
- If Reset/Clear selection in the previous step doesn't load correct data then click on reset.
Slicers
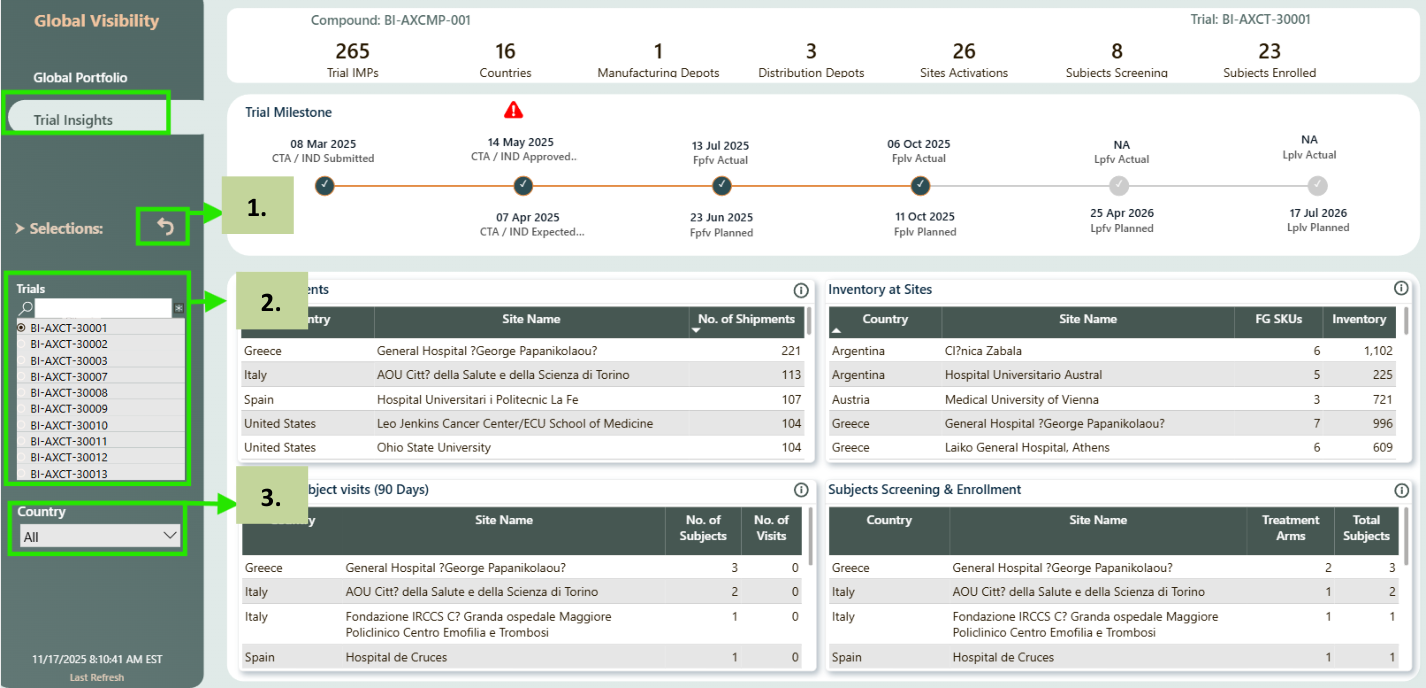
- Reset/Clear selection - Used to reset/clear all the above selected slicer values.
- Trials - Is single select and filters all visuals.
- Country - Is multiselect and filters all visuals.
Milestone details
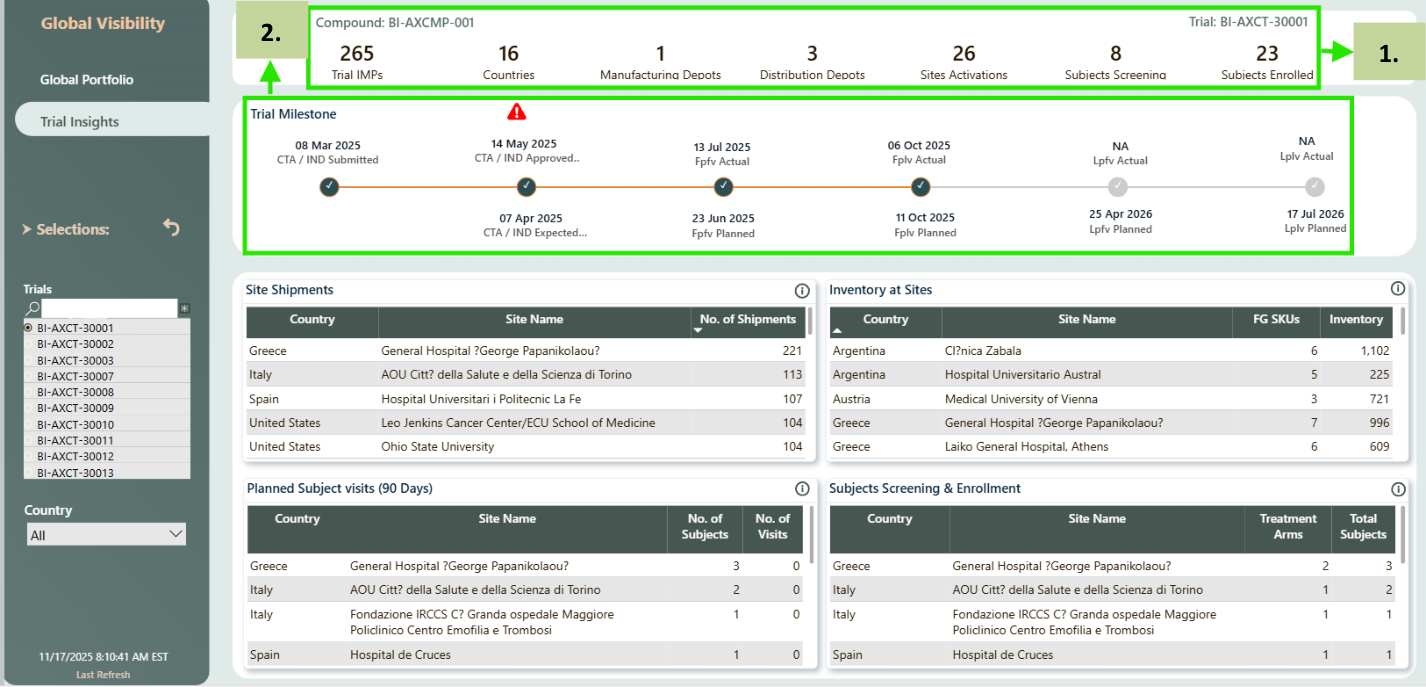
- This section provides a high-level summary of the selected trial, and all metrics update automatically based on the trial selected in the left-side Trials filter.
- These visual displays key milestones of the selected trial, it includes both planned and actual milestone dates. Any milestone that has not yet occurred will be marked as NA, while completed events display a checkmark indicator.
Details table
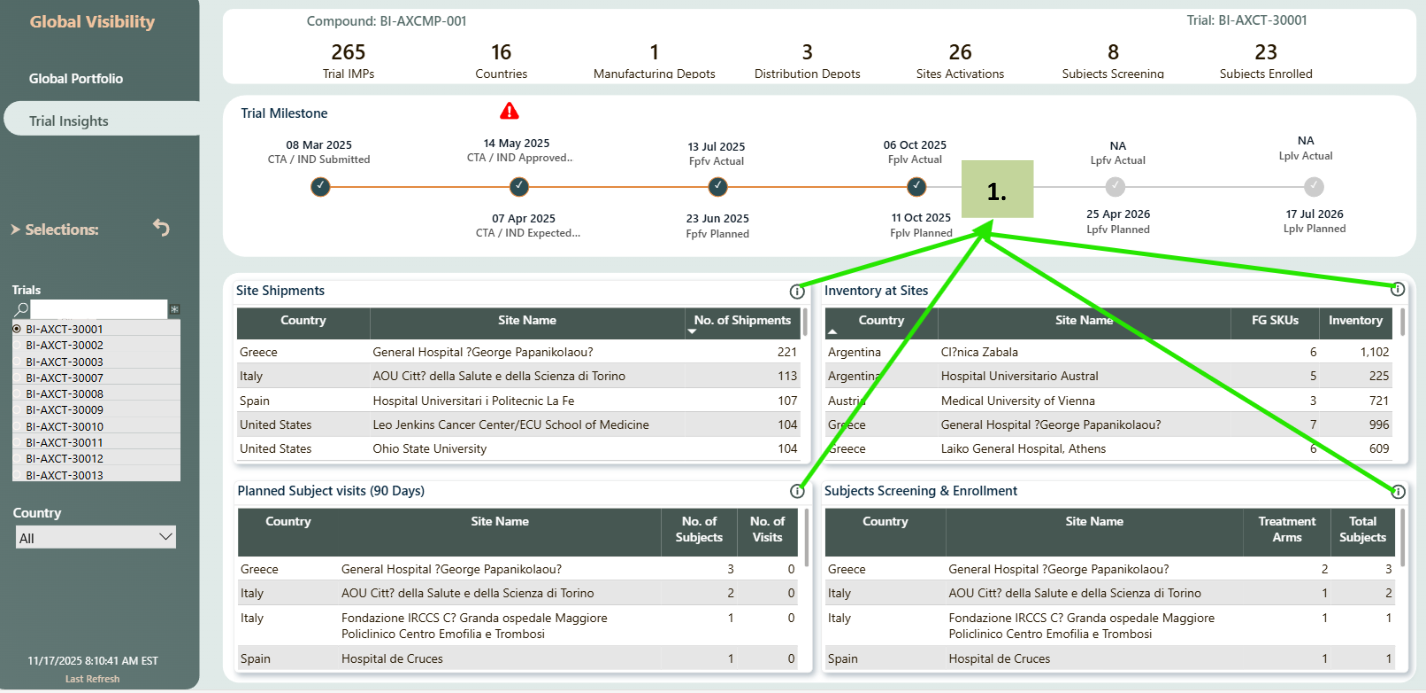
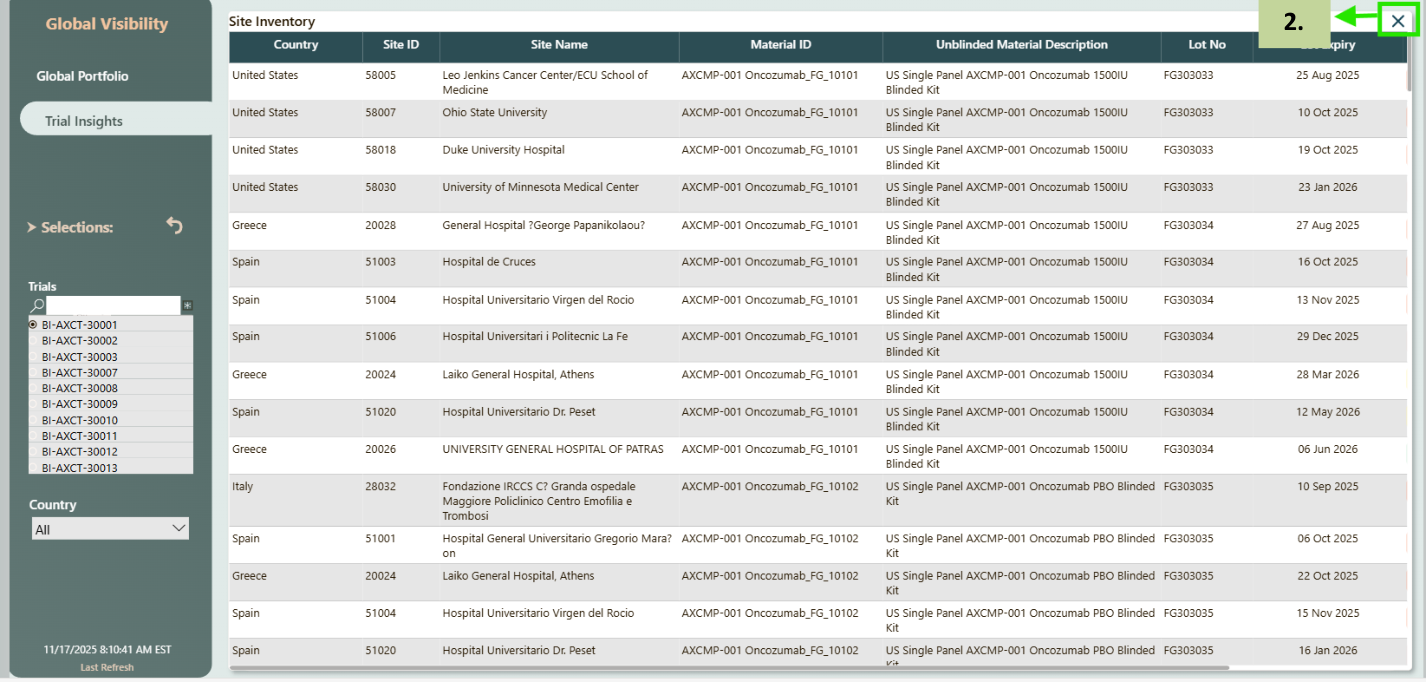
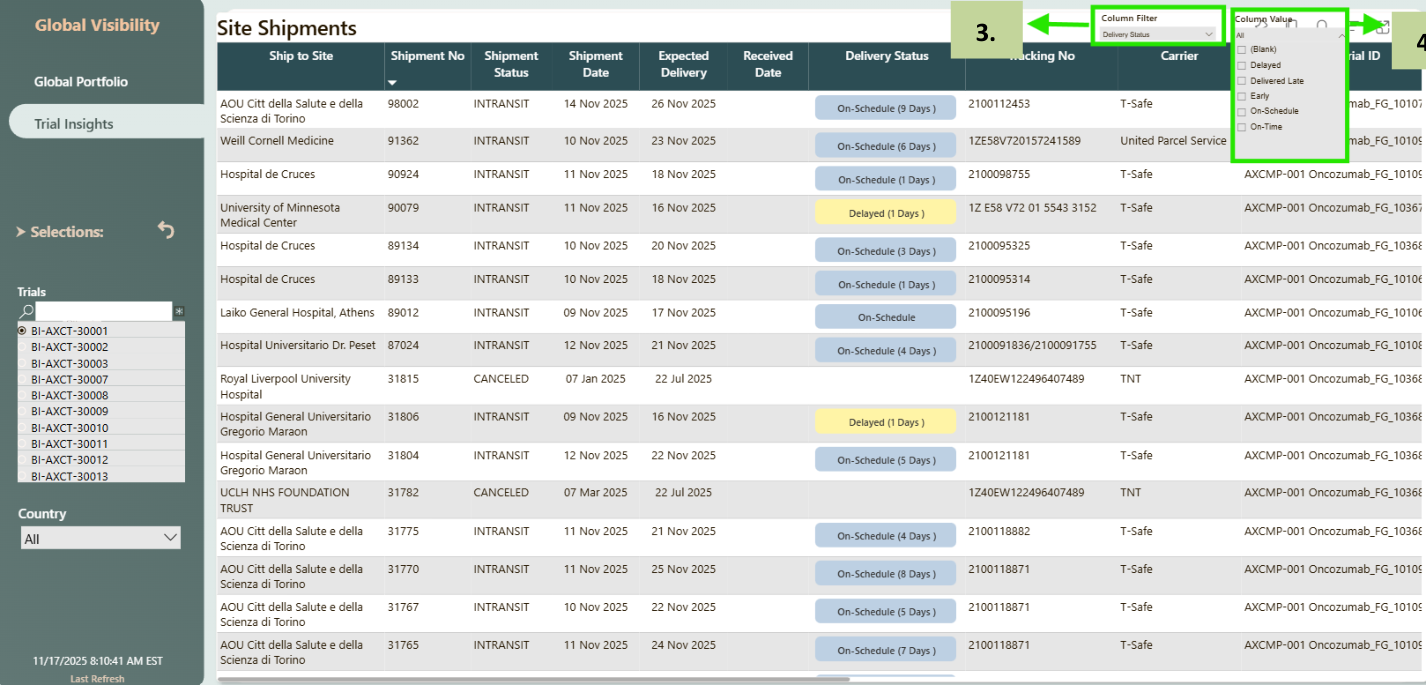
- If you click on the 'i' in any one of the tables (Site Shipments, Inventory At Sites, Upcoming Planned Visits (90 Days) or Subjects Screening & Enrollment) popup window will show up details table. In the pic given it is navigating to details for Site Shipments.
- Click on the 'X' on top right corner to close the popup window.
- The Column Filter dropdown allows you to choose which column you want to filter - either Shipment Status or Delivery Status.
- After selecting the desired column, the Column Values list displays all available values for that field. You can then select a value based on requirement, and the table will update to show only the matching records.
.db400dda.png&w=640&q=75)